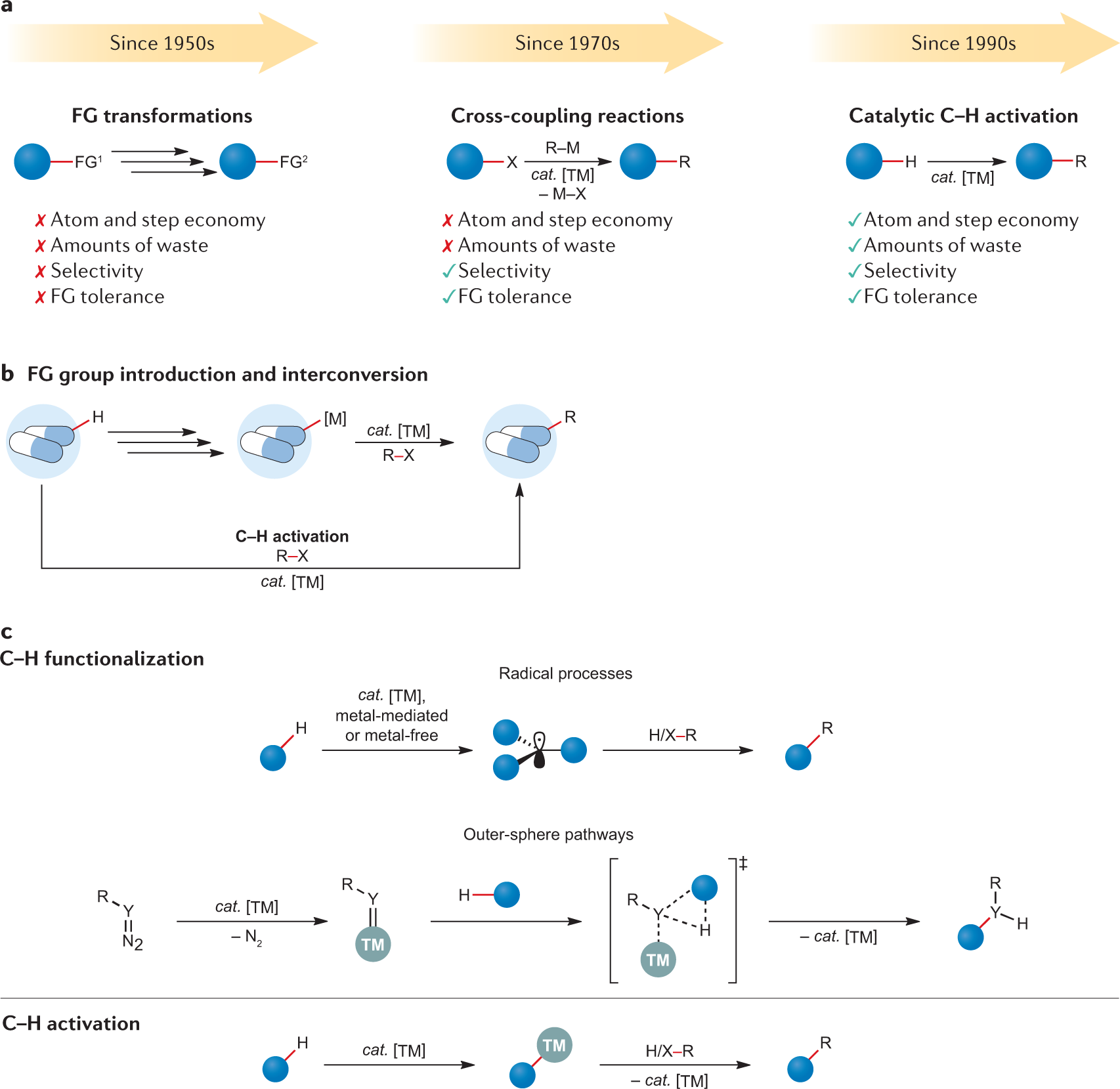
Palladium–Protein Oxidative Addition Complexes by Amine-Selective Acylation | Journal of the American Chemical Society

Palladium‐Catalyzed C–S Bond Formation: Rate and Mechanism of the Coupling of Aryl or Vinyl Halides with a Thiol Derived from a Cysteine - Moreau - 2005 - European Journal of Organic Chemistry -

On the Mechanism of Palladium-Catalyzed Aromatic C−H Oxidation | Journal of the American Chemical Society

Palladium-catalyzed oxidative arene C–H alkenylation reactions involving olefins: Trends in Chemistry

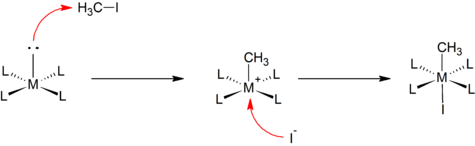
Inorganics | Free Full-Text | Structural Study of Model Rhodium(I) Carbonylation Catalysts Activated by Indole-2-/Indoline-2-Carboxylate Bidentate Ligands and Kinetics of Iodomethane Oxidative Addition

C-H Bond Activation via Concerted Metalation-Deprotonation at a Palladium(III) Center | Organometallic Chemistry | ChemRxiv | Cambridge Open Engage

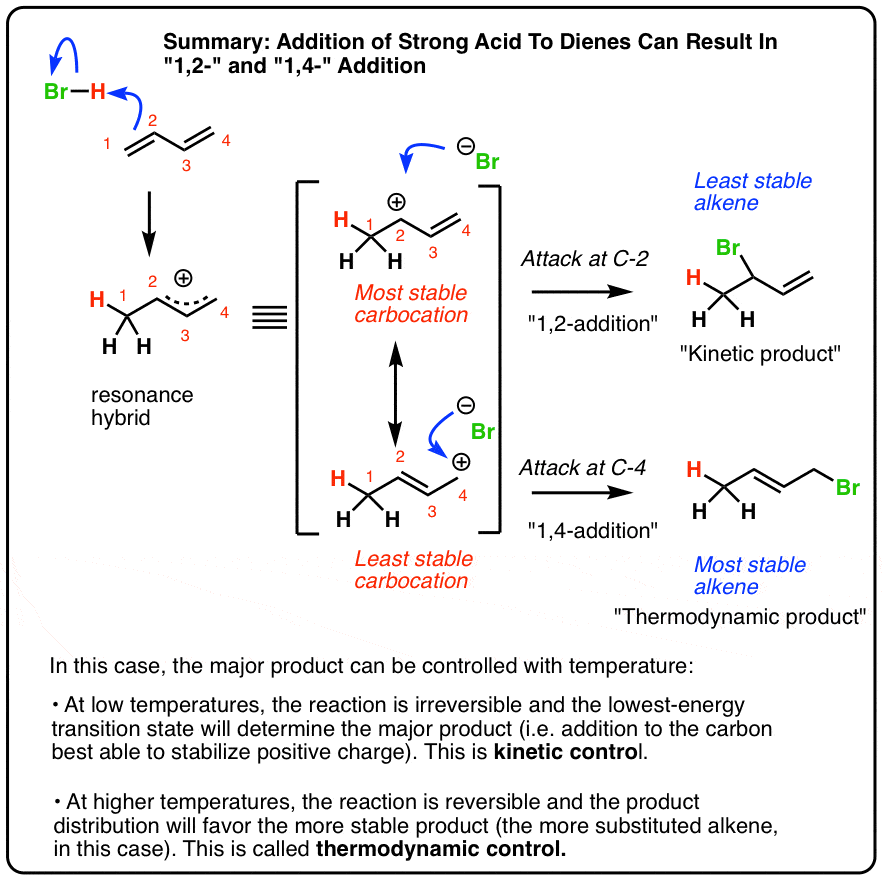
A Tutorial on Selectivity Determination in C(sp2)–H Oxidative Addition of Arenes by Transition Metal Complexes - ScienceDirect
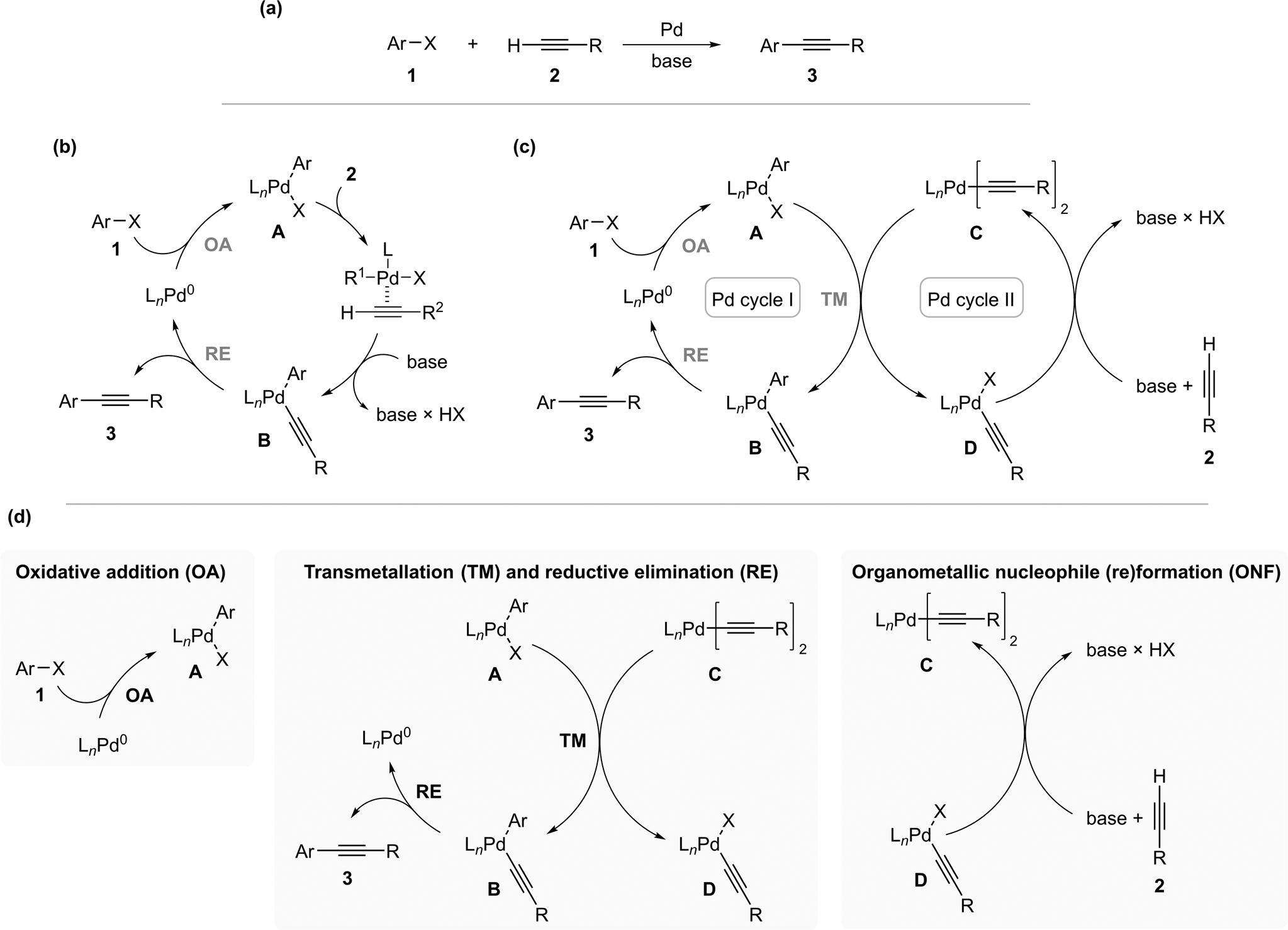
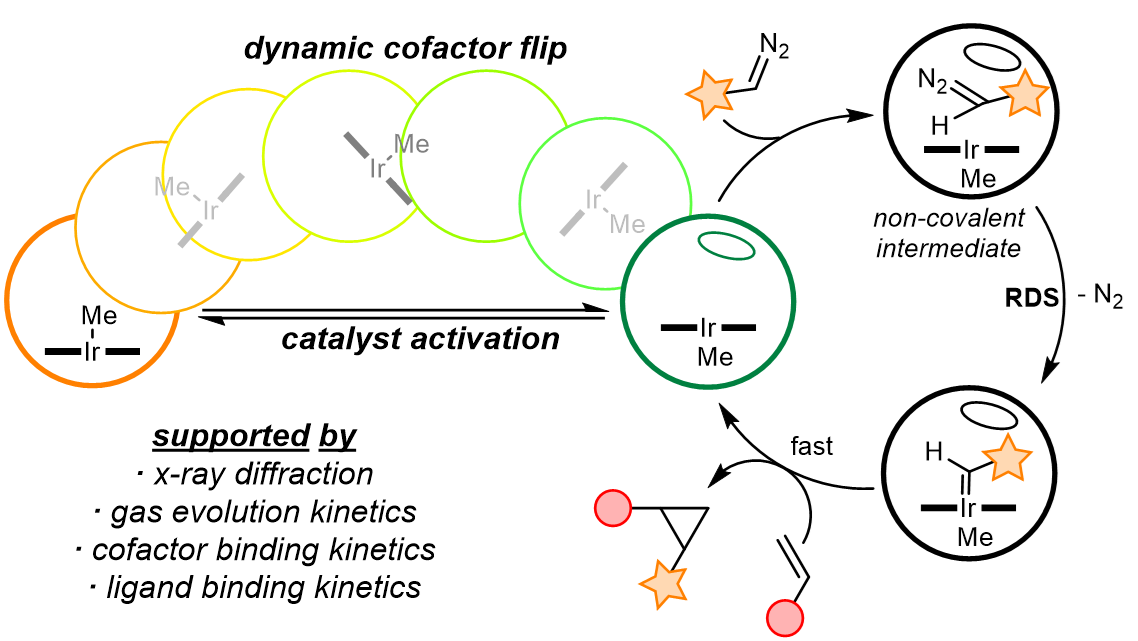
Elucidating the reaction mechanism of a palladium-palladium dual catalytic process through kinetic studies of proposed elementary steps | Communications Chemistry

PDF) Elucidating the reaction mechanism of a palladium-palladium dual catalytic process through kinetic studies of proposed elementary steps

Palladium(II)-catalyzed C-H activation/C-C cross-coupling reactions: versatility and practicality. - Abstract - Europe PMC

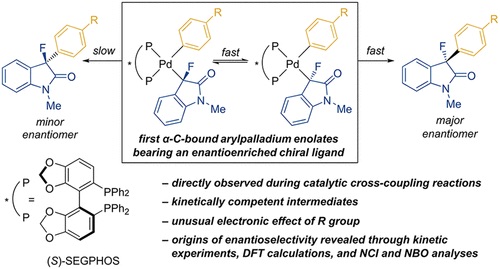
Kinetic Resolution of Benzylamines via Palladium(II)-Catalyzed C–H Cross-Coupling | Journal of the American Chemical Society
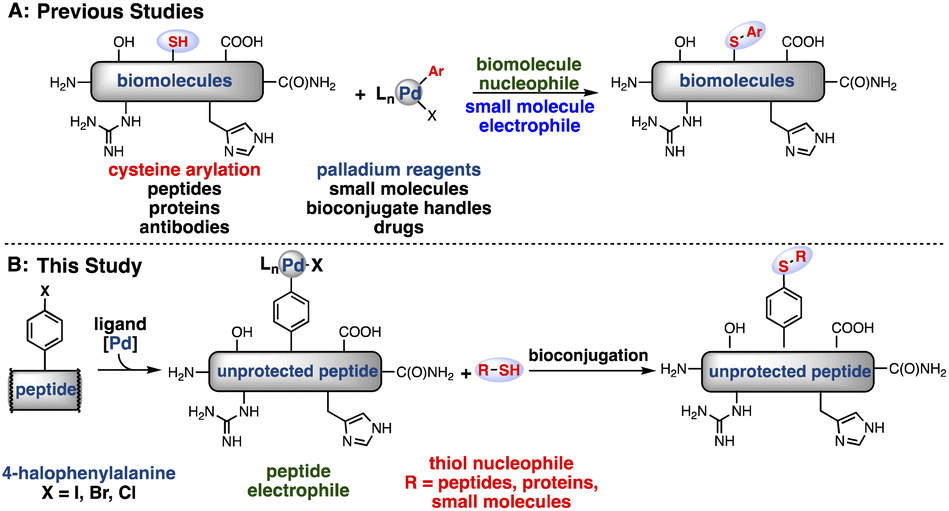
Palladium–peptide oxidative addition complexes for bioconjugation - Chemical Science (RSC Publishing) DOI:10.1039/D2SC04074C

Dynamic Kinetic Resolution of Biaryl Lactones via a Chiral Bifunctional Amine Thiourea-Catalyzed Highly Atropo-enantioselective Transesterification. | Semantic Scholar

Arylic C–X Bond Activation by Palladium Catalysts: Activation Strain Analyses of Reactivity Trends | Scientific Reports
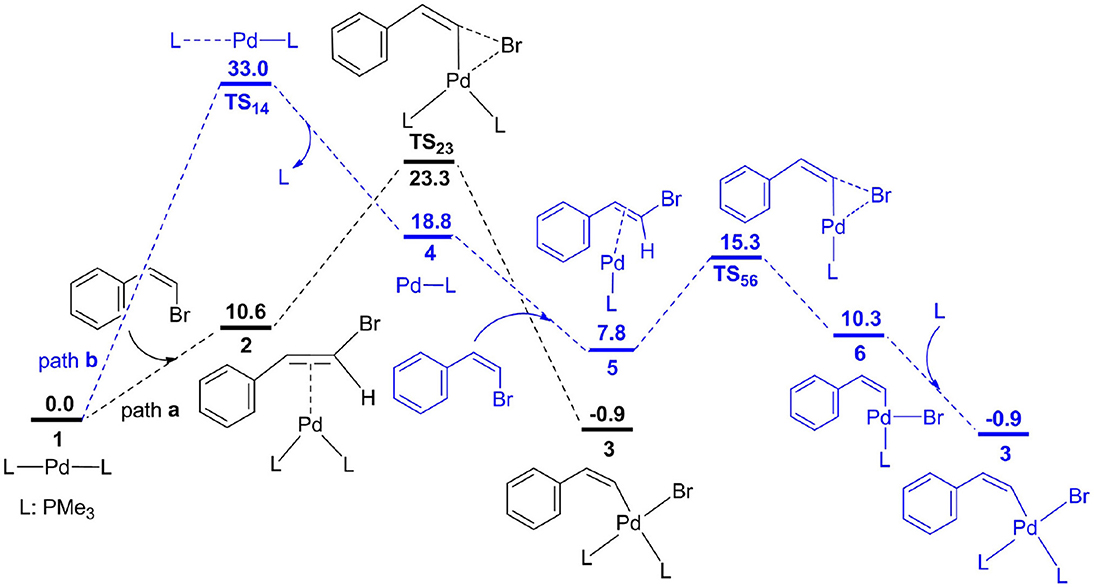
Frontiers | Key Mechanistic Features in Palladium-Catalyzed Methylcyclopropanation of Norbornenes With Vinyl Bromides: Insights From DFT Calculations

Inorganics | Free Full-Text | Structural Study of Model Rhodium(I) Carbonylation Catalysts Activated by Indole-2-/Indoline-2-Carboxylate Bidentate Ligands and Kinetics of Iodomethane Oxidative Addition